- Published on
What are cyborg cells? How scientists are merging living cells with electronics and nanomaterials
- Authors

- Name
- BioTech Bench
In 2015, a team at Harvard injected a mesh of silicon nanowires into living neurons and recorded electrical activity from inside the cells — for months. The cells survived. The electronics kept working. That experiment wasn't a one-off stunt. It was an early demonstration of something researchers have been building toward ever since: cells that don't just tolerate synthetic materials but integrate with them. Scientists now engineer cells that interface with nanomaterials, electronic components, and programmable gene circuits. The boundary between biological and artificial is no longer fixed.
This post is your map to what cyborg cells actually are, how they work, and why bench biologists are starting to encounter them.
What you'll learn in this post
- What makes a cell "cyborg" — the specific structural and functional criteria that separate this from ordinary cell biology
- The core engineering challenge at the bio-electronic interface and the main strategies researchers use to solve it
- The four main types of cyborg cell systems, with real experimental examples for each
- Where bench biologists actually run into this work — in assay design, drug testing, neural recording, and synthetic gene circuits
What is a cyborg cell?
Cyborg cells sit between fully biological living cells and fully synthetic systems — they are living cells functionally integrated with non-biological components. It means a biological entity that cannot be fully described by biology alone.
The key phrase is "functional integration." A cell with a nanoparticle stuck to its membrane from a failed transfection protocol is not a cyborg cell. A cell where a magnetic nanoparticle coating lets researchers steer it to a target site, load it with cargo, or trigger its behavior with an external field — that is a cyborg cell. The non-biological component changes what the cell can do.
The term became common in bioelectronics and synthetic biology during the 2010s. The 2023 paper by Contreras-Llano et al., which engineered bacteria with internal hydrogel skeletons that could survive conditions lethal to normal cells, brought "cyborg cell" firmly into mainstream use.
Four main design categories have emerged from the literature: cells with intracellular electronics or synthetic scaffolds, cells with surface-engineered non-biological coatings, cells carrying programmable synthetic gene circuits, and organoids embedded in electronic scaffolds. The next sections look at why building any of these is harder than it sounds, and then work through each category in turn.
The challenge: biology and electronics don't mix easily
Biological tissue and conventional electronics exist at opposite ends of nearly every physical scale that matters. Living tissue is soft — stiffness in the range of 0.1 to 10 kPa for most cells and brain matter — hydrated, electrochemically active, metabolically alive, and constantly remodeling its local environment. Silicon, the substrate most electronics are built on, has a stiffness closer to 100 GPa: roughly ten million times stiffer. That mechanical mismatch is not a minor inconvenience. When a rigid implanted electrode sits inside soft neural tissue, the immune system treats it as a persistent threat. Astrocytes and microglia encapsulate it in scar tissue, isolating it from the cells it was supposed to record from, and signal quality degrades within weeks of implantation.
Researchers have converged on four main strategies to bridge this gap. The first is flexible electronics: electrode arrays fabricated from thin-film polymers — parylene, SU-8, polyimide — instead of silicon, shaped into mesh or ribbon geometries that conform to tissue topology and flex with movement rather than fighting it. The second is conductive nanomaterials: carbon nanotubes, graphene flakes, and gold nanoparticles operate at the nanoscale where the stiffness mismatch with individual cell membranes is far smaller and surface-to-volume ratios are high enough to enable direct electrochemical coupling. The third is biocompatible conductive coatings: PEDOT:PSS, a water-swellable conductive polymer, is applied over electrode surfaces to make them hydrophilic and mechanically compliant at the tissue interface. The fourth is hydrogel carriers: soft crosslinked polymer networks — polyethylene glycol, alginate, gelatin methacryloyl — that embed electronic components while matching tissue mechanics in the 1 to 10 kPa range and allowing nutrient diffusion.
Solving the interface problem — mechanically, chemically, and electrically — is the engineering foundation all four types of cyborg cell systems build on.
Figure 1 shows a practical example of the conductive nanomaterials strategy: attaching nanoparticles to a microbial cell turns it into a biohybrid microrobot with new capabilities.
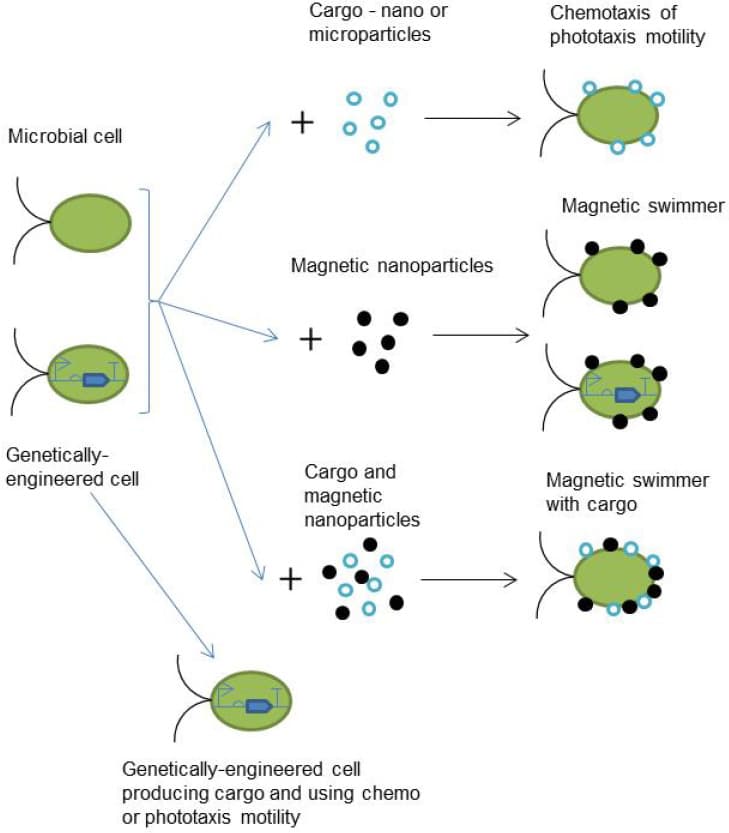
Figure 1. Building a biohybrid cell: a plain or genetically engineered microbial cell is paired with cargo nanoparticles, magnetic nanoparticles, or both to create biohybrid microrobots capable of chemotaxis, magnetic steering, or autonomous cargo delivery. Adapted from Gotovtsev P (2023). Microbial Cells as Microrobots: From Drug Delivery to Advanced Biosensors. _Biomimetics, 8(1):109. doi:10.3390/biomimetics8010109, under CC BY 4.0._
Four types of cyborg cells
Cyborg cell research has converged on four distinct design architectures, each solving the biology-electronics interface in a different way.
Intracellular nanowires
Researchers have demonstrated that ultra-fine silicon nanowires — some kinked into probe geometries — can be inserted directly into single cells or fashioned into three-dimensional scaffolds that cells actively engulf during culture. The Lieber group at Harvard pioneered this approach, with key demonstrations including nanowire tissue scaffolds (Tian et al. 2012, Nature Materials) and syringe-injectable mesh electronics small enough to be threaded through a needle and then unfurl inside tissue (Liu et al. 2015, Nature Nanotechnology). What these systems enable is continuous, long-duration recording of intracellular electrical activity without killing the cell — something traditional patch-clamp achieves acutely but cannot sustain over days or weeks without membrane disruption. Early demonstrations show stable recordings lasting months in cultured neurons, and if you work in neurophysiology or with stem-cell-derived cardiomyocytes, this class of tool is beginning to appear at the edge of the commercial recording landscape.
Surface-functionalized cells
A second approach leaves the cell's interior alone and instead engineers the outer surface, coating cells with nanoparticles — gold, iron oxide, or semiconductor quantum dots — or wrapping them in graphene oxide sheets. Attachment can be achieved covalently through click chemistry or metabolic labeling, or non-covalently through electrostatic interaction between the nanoparticle coating and the charged cell membrane. Each nanoparticle type brings a different capability: gold nanoparticles absorb near-infrared light and convert it to heat, enabling photothermal ablation of tumor cells from outside the body; iron oxide nanoparticles render cells visible by MRI and allow magnetic steering; cargo-loaded nanoparticles can ferry therapeutic payloads and release them at target sites. The central limitation of surface functionalization is coating stability across cell division — when a functionalized cell divides, daughter cells inherit progressively less of the original coating, so applications that require sustained function must account for dilution over multiple generations.
Genetically wired cells
The purest synthetic biology approach treats the cell's gene regulatory machinery as a programmable circuit board. Researchers design synthetic gene circuits by wiring transcription factors and promoters together in configurations that mirror electronic logic: an AND gate releases its protein output only when two distinct input signals are simultaneously present; a toggle switch holds a bistable state, acting as molecular memory; a genetic oscillator produces periodic pulses of output with no external clock. The founding demonstration is the Gardner, Cantor and Collins toggle switch published in E. coli in 2000 (Nature), the first bistable synthetic gene circuit and the paper that established a vocabulary of circuit design for the field. Modern implementations have moved into mammalian therapeutic cells: synthetic AND gates have been used to engineer T cells or stem cells that release a therapeutic payload only when two cancer-associated surface antigens are co-detected, reducing off-target activity. The connection to molecular biology is direct — dCas9-based gene regulation extends this circuit logic to programmable transcriptional control without permanent edits to the genome, meaning skills already common in CRISPR labs are directly transferable to synthetic circuit design.
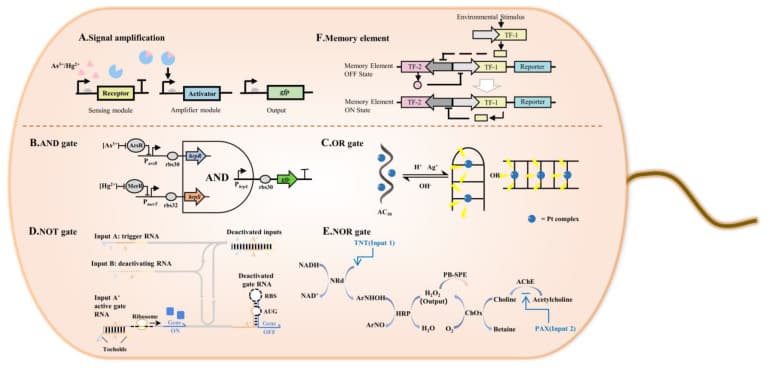
Figure 2. Gene circuit logic inside a living cell. Synthetic gene circuits can implement signal amplification (A), Boolean AND gates (B), OR gates (C), NOT gates via RNA toeholds (D), NOR gates via enzymatic cascades (E), and bistable memory elements (F). Adapted from Chen S et al. (2023). Advances in Synthetic-Biology-Based Whole-Cell Biosensors. _Int J Mol Sci, 24(9):7989. doi:10.3390/ijms24097989, under CC BY 4.0._
Hybrid organoids
Brain and cardiac organoids grown directly on or around multi-electrode arrays (MEAs) represent the most structurally complex cyborg cell system, allowing simultaneous recording and stimulation across thousands of electrodes in a three-dimensional tissue context that is substantially closer to in vivo physiology than conventional 2D cell culture on a flat electrode surface. Two landmark demonstrations bracketed the recent acceleration of this field: the DishBrain system reported by Kagan et al. in 2022 (Neuron) showed that cortical neurons plated on an MEA could learn to play Pong through electrophysiological feedback, demonstrating activity-dependent plasticity in a hybrid biological-electronic loop, and the organoid intelligence roadmap published by Smirnova et al. in 2023 laid out a systematic case for using brain organoids as biological computing substrates. For bench biologists, the practical entry point is already here — MEA plates from Axion BioSystems (Maestro) and Maxwell Biosystems are increasingly standard instruments in neurotoxicology labs, iPSC-derived cardiac organoid screening programs, and disease-modeling pipelines where network-level electrophysiology provides readouts that calcium imaging or patch-clamp alone cannot deliver.
What cyborg cells can do
Genetically wired bacteria or mammalian cells can detect specific molecules in their environment — arsenic, mercury, glucose, inflammatory cytokines, pathogen surface antigens — and produce a reporter signal: fluorescence, luminescence, or even a therapeutic drug. The key advantage over conventional biosensors is that the cell amplifies the signal, self-regenerates, and can be programmed to respond only when multiple inputs are simultaneously present, implementing AND logic that dramatically reduces false positives. The most-cited early example is the arsenic-detecting bacteria developed through successive iGEM projects and now being refined toward field-deployable water-quality monitoring.
Nanoparticle-loaded red blood cells and macrophages serve as stealthy drug carriers — the host cell membrane camouflages the payload from immune surveillance while the passenger nanoparticles carry the therapeutic cargo. Release can be triggered on demand by near-infrared light (gold NPs), an applied magnetic field (iron oxide NPs), or enzymatic signals from the tumor microenvironment. This targeting precision and immune evasion is not achievable with free nanoparticles injected directly into circulation.
Flexible mesh electronics and intracellular nanowires allow chronic recording and stimulation of neurons with minimal inflammatory scarring, because their mechanical compliance removes the stiffness mismatch that causes conventional electrodes to be encapsulated in scar tissue. Applications already in development include closed-loop brain-computer interfaces that adjust stimulation in real time, higher-channel-count cochlear implants, and disease-model platforms for studying neurodegeneration in Parkinson's and ALS models where network-level dynamics matter.
Cells running Boolean gene circuits can in principle perform computation — evaluating whether multiple conditions are simultaneously true and executing a programmed therapeutic or reporter response. The DishBrain experiment (Kagan et al. 2022, Neuron) showed that cortical neurons on an MEA modified their firing patterns in response to Pong game feedback, demonstrating activity-dependent plasticity in a biological-electronic loop. This is still basic research rather than deployed technology, but it established that bidirectional communication between living neural networks and electronic systems is achievable outside a living organism.
Engineered bacteria with biosensor circuits are being tested for detecting and flagging heavy metal contamination in soil and water; some strains combine the sensing function with active metal sequestration, linking detection to remediation within a single cell and removing the need for separate indicator and cleanup steps.
Why bench biologists are starting to encounter this
If you work with organoids, iPSC-derived neurons, or cardiomyocytes, MEA recording is increasingly standard infrastructure — you may already be running experiments on a Maestro or Maxwell plate without thinking of them as cyborg cell systems. The cyborg organoid literature directly informs how to interpret network-level electrophysiology data from these instruments, because the same principles of bio-electronic coupling that govern signal fidelity in research MEA systems also govern what those firing patterns actually mean about tissue state.
If you work with CRISPR, synthetic gene circuits are the natural extension of programmable gene regulation. CRISPRi and CRISPRa are already functional gene circuit components — they are transcriptional inputs that can be wired into AND gates, toggle switches, and feedback loops. Learning the logic gate vocabulary opens a design space that goes well beyond single-gene knockouts and knockins, toward cells that sense a combination of conditions, compute a response, and execute it autonomously.
If you use lipid nanoparticles or gold nanoparticles for transfection or delivery experiments, you are already working with hybrid cell technology. The line between a transfection vehicle and a surface-functionalized cyborg cell is a question of design intent, not a categorical difference in the underlying chemistry.
The field is moving fast enough that tools now in research labs are likely to appear in core facilities within five years.
Where the field is going
Four challenges dominate current research and engineering effort. Longevity is unsolved at scale: electronic implants tend to outlast cells, while cells divide and progressively dilute nanoparticle coatings and synthetic circuit components across daughter generations, making sustained function difficult to guarantee. Scalability remains a bottleneck because most cyborg cell fabrication techniques — nanowire injection, surface functionalization with controlled orientation, hydrogel encapsulation of electrode arrays — require specialized deposition equipment or slow manual assembly that does not translate easily to the batch sizes needed for clinical or commercial use. Immune response to non-biological components is a persistent problem: PEDOT:PSS coatings and hydrogel encapsulants reduce acute foreign-body reactions, but long-term in vivo function is still difficult to achieve because chronic low-grade inflammation gradually degrades both the biological and electronic components. Ethics is an active conversation: implanting electronic tissue interfaces that blur the boundary between person and device raises questions about identity, consent, and long-term reversibility that the field is beginning to address through dedicated bioethics frameworks, but regulatory and philosophical consensus is well behind the technical capability.
Near-term milestones are already visible. Organoid-on-chip systems for drug toxicity testing are entering commercial and regulatory pipelines — some organ chip models have received FDA qualification as drug development tools, replacing certain animal testing steps. Closed-loop neural prosthetics based on flexible mesh electronics are in clinical trials for sensory restoration, with cochlear and retinal implant programs leading and cortical BCI systems not far behind. Therapeutic cells with synthetic AND gate logic — specifically CAR-T cells engineered with safety switch circuits that require co-detection of two tumor antigens before activating — are entering early clinical testing, translating gene circuit design directly into oncology. For a bench biologist, the practical implication is that understanding gene circuits, MEA readouts, and nanoparticle-cell interactions is increasingly useful knowledge, not niche specialization.
Resources and further reading
Key papers, reviews, and tools for going deeper.
| Resource | What it covers |
|---|---|
| Gardner TS, Cantor CR, Collins JJ (2000). Construction of a genetic toggle switch in Escherichia coli. Nature, 403:339–342. | Foundational synthetic biology paper: the first bistable gene circuit built in a living cell — the conceptual seed of all genetically wired cyborg cells. |
| Tian B et al. (2012). Macroporous nanowire nanoelectronic scaffolds for synthetic tissues. Nature Materials, 11:986–994. | Landmark Lieber lab paper demonstrating 3D nanowire scaffolds seeded with neurons and cardiomyocytes — the first cyborg tissue where electronics are woven into living tissue. |
| Liu J et al. (2015). Syringe-injectable electronics. Nature Nanotechnology, 10:629–636. | Demonstrates injection of flexible mesh electronics into the brain via syringe; the mesh unfurls inside tissue and records neurons chronically for months. |
| Contreras-Llano LE et al. (2023). Engineering Cyborg Bacteria Through Intracellular Hydrogelation. Advanced Science, 10(9):e2204175. | The paper that coined "cyborg cells" by name: bacteria with an internal hydrogel skeleton that preserves metabolism and gene circuit function while gaining new stress resistance. |
| Chen S et al. (2023). Advances in Synthetic-Biology-Based Whole-Cell Biosensors: Principles, Genetic Modules, and Applications in Food Safety. Int J Mol Sci, 24(9):7989. | Accessible review of how AND gates, OR gates, NOT gates, and memory elements are wired into bacterial cells to create programmable living sensors. |
| Teixeira AP, Fussenegger M (2024). Synthetic Gene Circuits for Regulation of Next-Generation Cell-Based Therapeutics. Advanced Science, 11(8):e2309088. | Review of gene circuit designs for therapeutic mammalian cells — covers CAR-T logic gates, designer receptors, and light-inducible circuits. |
| Gotovtsev P (2023). Microbial Cells as Microrobots: From Drug Delivery to Advanced Biosensors. Biomimetics, 8(1):109. | Overview of microbial cell biohybrids: how attaching nanoparticles to cells creates microrobots with chemotaxis, magnetic steering, and autonomous cargo delivery. |
| Kagan BJ et al. (2022). In vitro neurons learn and exhibit sentience when embodied in a simulated game-world. Neuron, 110(23):3952–3969. | DishBrain: human and mouse cortical neurons on a high-density MEA learned to play Pong via electrophysiological feedback — a landmark paper for hybrid organoid computing. |
| Smirnova L et al. (2023). Organoid intelligence (OI): the new frontier in biocomputing and intelligence-in-a-dish. Frontiers in Science, 1:1017235. | Roadmap for organoid intelligence: brain organoids + MEA arrays as biological processors, with scalable culturing approaches and an ethical framework. |
| Jang S et al. (2021). Biomaterials for Cell-Surface Engineering and Their Efficacy. J Funct Biomater, 12:41. | Review of polymer and biomaterial coating strategies for therapeutic mammalian cells — layer-by-layer assembly, cell encapsulation, and immune protection for surface-functionalized cyborg cells. |